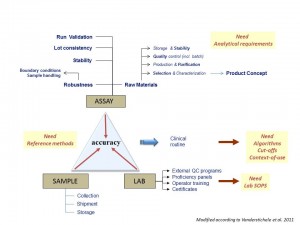
BIOMARKABLE guarantees you that an accurate quantification of your proteins in biological samples will be obtained after extensive development and standardization at the level of the ASSAY, the SAMPLE and the LABORATORY.
BIOMARKABLE can consult in
- all or specific parts of the development of your protein assays
- (cost efficient) assay development, including experimental designs with statistical power
- the generation, selection and quality control for critical raw materials or biological samples
- the preparation of a plan for Quality Control, Process qualification, or clinical evaluation.
- the initiation and follow-up of stability studies
- the transfer of your product from single analyte to multi-plex technologies
BIOMARKABLE can provide daily follow-up services, as well as the permanent reporting line of project progression, compliant with regulatory requirements.
The skills of the individuals in the team will be optimally integrated in the development plan.
Once available in the market, BIOMARKABLE can assist in Product Life Cycle Management: assay comparison, translation to other technology platforms, broaden application area (= fit-for-purpose), problem solving, creation of network.